

38-year-old male

Taxi driver

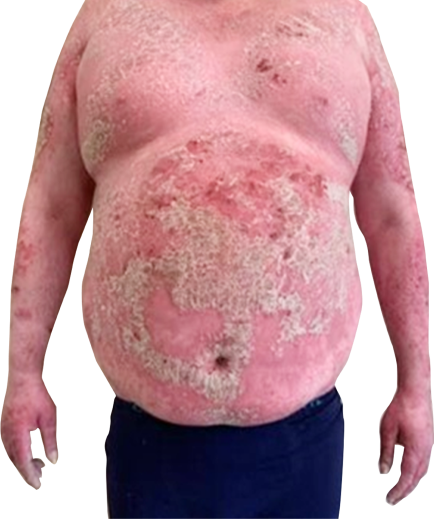
Severe plaque-type psoriasis

Not achieving treatment goal - clear/almost clear skin
Watch Prof. Torres talk about the case and provide the rationale behind the recommendations presented
Skyrizi is indicated for the treatment of moderate to severe plaque psoriasis in adults who are candidates for systemic therapy.1
Deidentified patient case provided by Prof. Tiago Torres, Instituto de Ciências Biomédicas Abel Salazar, University of Porto, Porto, Portugal, with patient’s consent. For illustrative purposes only, individual results will vary depending on patient’s unique situation. Pseudonyms have been used to protect patient anonymity. Case study is a clinical observation of a single patient experience. AbbVie makes no representation or promise of similar results in any patient.
1. SKYRIZI (risankizumab) Summary of Product Characteristics. AbbVie Ltd; September 2023. This promotional website is organized by AbbVie and is for Healthcare professionals only.

15-year diagnosed psoriasis disease duration

No symptoms of PsA

Current presentation:
- BSA >70%, PASI 42.3, DLQI 19

Severe plaque psoriasis

Previous treatments include:
- Cyclosporine - stopped due to an adverse event
- MTX - inadequate response
- TNFi (adalimumab) - 40 mg every other week maintenance dosing (after starter doses) for about 2 years; inadequate response

No treatments currently
Absolute PASI
42.3
BSA
>70%
DLQI
19
BMI, body mass index; BSA, body surface area; DLQI, Dermatology Life Quality Index; HDL, high-density lipoprotein; LDL, low-density lipoprotein; MTX, methotrexate; PASI, Psoriasis Area and Severity Index; PsA, psoriatic arthritis; TNFi, tumor necrosis factor inhibitor.
Deidentified patient case provided by Prof. Tiago Torres, Instituto de Ciências Biomédicas Abel Salazar, University of Porto, Porto, Portugal, with patient’s consent. For illustrative purposes only, individual results will vary depending on patient’s unique situation. Pseudonyms have been used to protect patient anonymity. Case study is a clinical observation of a single patient experience. AbbVie makes no representation or promise of similar results in any patient.

The patient is concerned about the reactions of his clients “My clients look at me with a strange face.”

Comorbidities:
Obesity (BMI 35 kg/m2)
Hyperlipidemia

Unhealthy lifestyle: no physical activity and high-calorie diet
BMI, body mass index; BSA, body surface area; DLQI, Dermatology Life Quality Index; HDL, high-density lipoprotein; LDL, low-density lipoprotein; MTX, methotrexate; PASI, Psoriasis Area and Severity Index; PsA, psoriatic arthritis; TNFi, tumor necrosis factor inhibitor.
Deidentified patient case provided by Prof. Tiago Torres, Instituto de Ciências Biomédicas Abel Salazar, University of Porto, Porto, Portugal, with patient’s consent. For illustrative purposes only, individual results will vary depending on patient’s unique situation. Pseudonyms have been used to protect patient anonymity. Case study is a clinical observation of a single patient experience. AbbVie makes no representation or promise of similar results in any patient.
What next?
What would be your treatment target with this patient?

‘Clear or almost clear’ skin has become a treatment target in many guidelines
BSA, body surface area; DLQI, Dermatology Life Quality Index; PASI, Psoriasis Area Severity Index; PGA, Physician’s Global Assessment; VAS, visual analogue scale.
1. Papp AK, et al. J Cutan Med Surg. 2011;15(4):210–219; 2. Armstrong A, et al. J Am Acad Dermatol. 2017;76(2):290–298; 3. Amatore F, et al. J Eur Acad Dermatol Venereol. 2019;33(3):464–483; 4. Smith CH, et al. Br J Dermatol. 2020;183(4):628–637; 5. Saeki H, et al. J Dermatol. 2020;47(3):201–222; 6. Echeverría C, et al. SOARPSO. http://www.soarpso.org/recursos/archivos/AG_psoriasis_2020_english.pdf [Last accessed June 2023]; 7. Nast A, et al. J Eur Acad Dermatol Venereol. 2020; 34(11):2461-2498; 8. Gomez M, et al. Acadiia Mexicana de Dermatologica. https://071d2186-dd73-483c-9a76-5f7e2b5bca70.filesusr.com/ugd/f187f1_651eb668a34b494f9a4be43a8b8773cc.pdf [Last accessed June 2023].
What next?
Which factors from Ahmed's presentation and latest clinical assessment would influence your management decisions?

EuroGuiDerm treatment algorithm for moderate-to-severe psoriasis1
Overview of main recommendations for initiation and selection of a systemic treatment


According to the EuroGuiDerm guidelines higher treatment goals should be the aim and include achieving PASI 90 or PASI 100. The focus has shifted towards a targeted final outcome which includes PASI ≤2, DLQI <2 or PGA clear or almost clear.1
‘Label’ as approved therapeutic indication by the European Medicines Agency.
*First line refers to the therapeutic indication as approved by the European Medicines Agency.
†Please refer to individual SmPCs for information on prescribing specific treatments.
csDMARD, conventional synthetic disease-modifying antirheumatic drug; DLQI, Disability Life Quality Index; IL, interleukins; PASI, Psoriasis Area and Severity Index; PDE, phosphodiesterase inhibitors; PGA, Physician Global Assessment; SmPC, summary of product characteristics; TNF, tumor necrosis factor.
1. Nast A, et al. Available at: https://www.guidelines.edf.one//uploads/attachments/cl27nt7yb001q90jnmykcah83-euroguiderm-pso-gl-feb-2022.pdf [Last accessed on: August 2023].
BSA, body surface area; PASI, Psoriasis Area and Severity Index; PGA, Physician’s Global Assessment.
1. Papp AK, et al. J Cutan Med Surg. 2011;15(4):210–219; 2. Armstrong A, et al. J Am Acad Dermatol. 2017;76(2):290–298; 3. Smith CH, et al. Br J Dermatol. 2020;183(4):628–637; 4. Saeki H, et al. J Dermatol. 2020;47(3):201–222; 5. Echeverría C, et al. SOARPSO. http://www.soarpso.org/recursos/archivos/AG_psoriasis_2020_english.pdf [Last accessed June 2023]; 6. Nast A, et al. https://www.guidelines.edf.one//uploads/attachments/cl27nt7yb001q90jnmykcah83-euroguiderm-pso-gl-feb-2022.pdf [Last accessed on: August 2023]; 7. Gomez M, et al. Academia Mexicana de Dermatologica. https://071d2186-dd73-483c-9a76-5f7e2b5bca70.filesusr.com/ugd/f187f1_651eb668a34b494f9a4be43a8b8773cc.pdf [Last accessed June 2023]; 8. SKYRIZI (risankizumab) Summary of Product Characteristics. AbbVie Ltd; September 2023.


A decision was made to start Ahmed on SKYRIZI (risankizumab) 150 mg at Weeks 0 and 4, then every 12 weeks thereafter.8
Skyrizi is indicated for the treatment of moderate to severe plaque psoriasis in adults who are candidates for systemic therapy.
Skyrizi is contraindicated in patients hypersensitive to the active substance or to any of the excipients, and in patients with clinically important active infections (e.g. active tuberculosis). Risankizumab may increase the risk of infection. In patients with a chronic infection, a history of recurrent infection, or known risk factors for infection, risankizumab should be used with caution. Treatment with risankizumab should not be initiated in patients with any clinically important active infection until the infection resolves or is adequately treated.
It is important to consider the following warnings and precautions before starting treatment with risankizumab:8
- Treatment with risankizumab should not be initiated in patients with any clinically important active infection until the infection resolves or is adequately treated
- Prior to initiating treatment with risankiziumab, patients should be evaluated for tuberculosis (TB) infection. Anti-TB therapy should be considered prior to initiating risankizumab in patients with a history of latent or active TB in whom an adequate course of treatment cannot be confirmed
- Prior to initiating therapy with risankizumab, completion of all appropriate immunizations should be considered according to current immunization practices. If a patient has received live vaccination, it is recommended to wait at least 4 weeks prior to starting treatment with risankizumab
‘Label’ as approved therapeutic indication by the EMA (European Medicines
Agency). *First line refers to the therapeutic indication as approved by the
European Medicines Agency.
†Please refer to individual SmPCs for information on prescribing
specific treatments.
Ahmed was started on SKYRIZI with every-12-week dosing* after initiation doses

SKYRIZI® (risankizumab) data
Click Next to see the SKYRIZI efficacy and safety data that informed this choice for Ahmed
SKYRIZI® (risankizumab) label indication and body weight guidance based on patient clinical profile1
- SKYRIZI® (risankizumab) is indicated for the treatment of moderate to severe plaque psoriasis in adults who are candidates for systemic therapy
- SKYRIZI® (risankizumab) clearance and volume of distribution increase as body weight increases which may result in reduced efficacy in subjects with high body weight (>130 kg). However, this observation is based on a limited number of subjects. No dose adjustment based on body weight is currently recommended
*SKYRIZI is dosed as 150 mg Q12W after initiation doses at Weeks 0 and 4.
Consideration should be given to discontinuing treatment in patients who
have shown no response after 16 weeks of treatment. Some plaque psoriasis
patients with initial partial response may subsequently improve with
continued treatment beyond 16 weeks.
Q12W, every 12 weeks.
Deidentified patient case provided by Prof. Tiago Torres, Instituto de Ciências Biomédicas Abel Salazar, University of Porto, Porto, Portugal, with patient’s consent. For illustrative purposes only, individual results will vary depending on patient’s unique situation. Pseudonyms have been used to protect patient anonymity. Case study is a clinical observation of a single patient experience. AbbVie makes no representation or promise of similar results in any patient.
1. SKYRIZI (risankizumab) Summary of Product Characteristics. AbbVie Ltd; September 2023.
IMMvent study design1
A 44-week, randomized comparative study versus adalimumab in adult patients with moderate to severe chronic plaque psoriasis (N=605)


*Administered as two 75 mg subcutaneous injections. SKYRIZI (risankizumab) 150 mg was dosed at Weeks 0 and 4 in part A. In part B, patients re-randomized to SKYRIZI (risankizumab) were given 150 mg at Weeks 16, 20, and 32; and patients remaining on SKYRIZI (risankizumab) were given study drug at Weeks 16 and 28.
** Adalimumab dosing was 80 mg at baseline, 40 mg at Week 1, and then 40 mg Q2W
PASI, Psoriasis Area and Severity Index; Q2W, every 2 weeks; SC, subcutaneous; sPGA, static Physician’s Global Assessment.
1. Reich K, et al. Lancet. 2019;394:576–586, and Supplementary appendix.
IMMvent patient demographics and baseline characteristics1


IMMvent data is from the intent-to-treat population in Part A, consisting of all randomized patients.
*Weight >100 kg and prior TNF exposure were stratification factors for randomization.†Patients with prior adalimumab use were excluded in IMMvent.
ADA, adalimumab; BSA, body surface area; PASI, Psoriasis Area and Severity Index; TNF, tumor necrosis factor.
1.Reich K, et al. Lancet. 2019;394:576–586.
IMMvent study Part A: SKYRIZI demonstrated superior complete skin clearance (PASI 90 and 100) versus adalimumab at Week 16 (NRI)1



Week 16 endpoints for PASI 90 (co-primary) and PASI 100 (ranked secondary) were multiplicity-controlled. All other data points shown were prespecified, non-ranked endpoints that were not controlled for multiplicity.
Co-primary endpoints for Part A – PASI 90 and sPGA 0 or 1 at Week 16, SKYRIZI versus adalimumab – were met (sPGA 0 or 1 data not shown), p<0.0001. *Nominal p=0.0012 versus adalimumab at Week 8 for PASI 90. †Nominal p<0.0001 versus adalimumab at Week 12 for PASI 90 and PASI 100. ‡Nominal p=0.0171 versus adalimumab at Week 8 for PASI 100. $p<0.0001 versus adalimumab at Week 16 for PASI 90 and PASI 100.
NRI, non-responder imputation; PASI, Psoriasis Area and Severity Index; sPGA, static Physician’s Global Assessment.
1. Reich K, et al. Lancet. 2019;394:576–586.
IMMvent study Part B: SKYRIZI demonstrated higher rates of skin clearance (PASI 90 and 100) versus adalimumab at Week 44 in re-randomized adalimumab patients with PASI≥50 to PASI<90 response at Week 16 (NRI)1
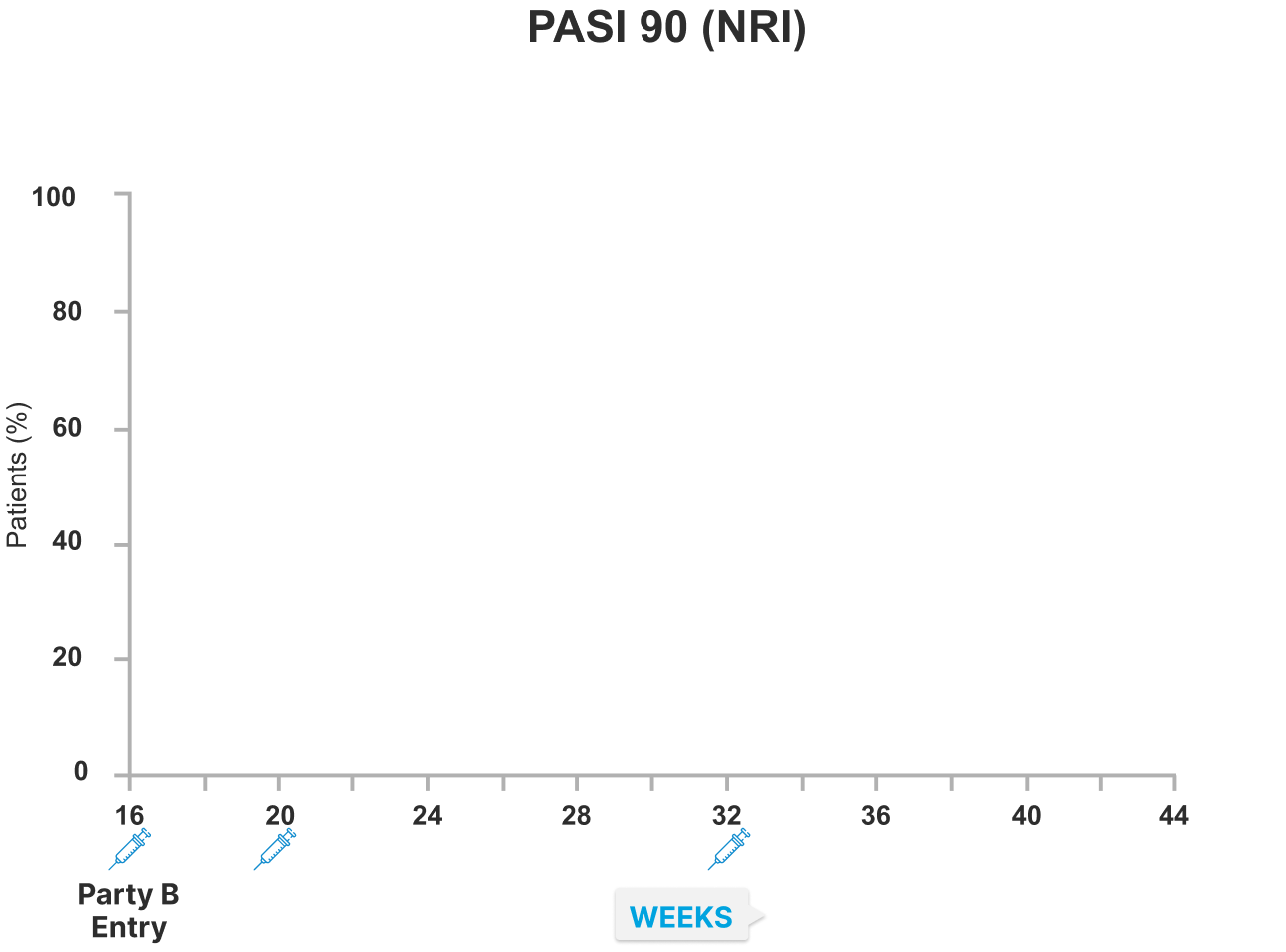

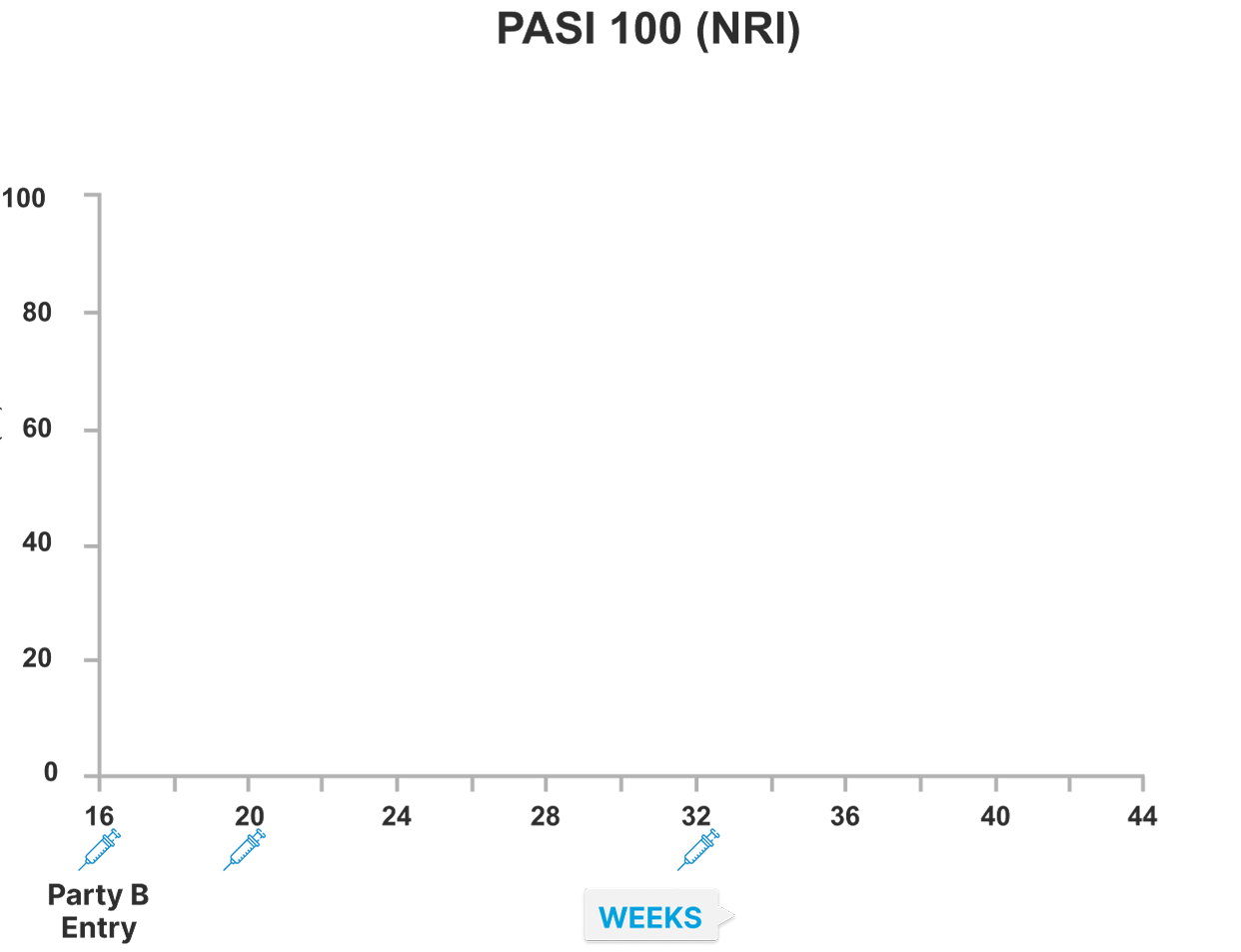


In part B re-randomized patients, Week 44 endpoints for PASI 90 (primary) and PASI 100 (ranked secondary) were multiplicity-controlled. All other data points shown were prespecified, non-ranked endpoints that were not controlled for multiplicity.
Patients achieving a ≥PASI 50 to <PASI 90 response with ADA at Week 16 were re-randomized to either SKYRIZI or ADA.
*Nominal p=0·0142 versus adalimumab at Week 20 for PASI 90. †Nominal p=0.0001 versus adalimumab at Week 24 for PASI 90. ‡Nominal p<0.0001 versus adalimumab at Weeks 28, 32, 36 and 40 for PASI 90 and at Week 36 for PASI 100. ¶Nominal p=0.0220 versus adalimumab at Week 24 for PASI 100. ||Nominal p=0.0022 versus adalimumab at Week 28 for PASI 100. **Nominal p=0.0073 versus adalimumab at Week 32 for PASI 100. §Nominal p=0.0002 versus adalimumab at Week 40 for PASI 100. $p<0.0001 versus adalimumab at Week 44 for PASI 90 and PASI 100.
ADA, adalimumab; NRI, non-responder imputation; PASI, Psoriasis Area and Severity Index.
1. Reich K, et al. Lancet. 2019;394:576–586.
SKYRIZI safety profile in Phase 3 IMMvent trial1
Overview of TEAEs reported in IMMvent Phase 3 trial across 605 patients in the first 16 weeks of treatment1
| IMMvent1 (N=605) | ||
|---|---|---|
| Events, % | SKYRIZI (n=301) | Adalimumab (n=304) |
| Any adverse event | 168 (56%) | 173 (57%) |
| Drug-related adverse event* | 55 (18%) | 61 (20%) |
| Serious adverse event | 10 (3%) | 9 (3%) |
| Drug-related serious adverse event* | 2 (1%) | 4 (1%) |
| Severe adverse event | 10 (3%) | 10 (3%) |
| Adverse event leading to drug discontinuation | 4 (1%) | 6 (2%) |
| Infection† | 88 (29%) | 74 (24%) |
| Serious infection | 1(<1%) | 1(<1%) |
| Opportunistic infection | 0 | 1(<1%) |
| Active tuberculosis | 0 | 0 |
| Latent tuberculosis | 0 | 0 |
| Serious hypersensitivity | 0 | 0 |
| Adjudicated major adverse cardiovascular event | 1(<1%)‡ | 0 |
| Hepatic events | 5 (2%) | 3 (1%) |
| Malignancy | 1(<1%) | 1(<1%) |
| Malignancy excluding non-melanoma skin cancer | 1(<1%)§ | 1(<1%)¶ |
| Deaths (including non-treatment emergent) | 1(<1%)‡ | 2(<1%)¶|| |
Data are n (%). Percentages are rounded.
*Investigator assessed adverse event as possibly related to study drug. †The most frequently reported infectious adverse events were viral upper respiratory tract infection and upper respiratory tract infection. ‡One patient with acute myocardial infarction on study day 73 (event was not considered to be study drug related by investigator). ¶One patient diagnosed with invasive lobular breast carcinoma on study day 63 following routine mammogram (event was not considered to be study drug related by investigator). ¶One patient with stage IV gall bladder cancer. ||One patient with cholelithiasis underwent gall bladder surgery, had cardiopulmonary arrest, and died due to abdominal abscess, sepsis, and gastric perforation (events were not considered to be study drug related by investigator).
TEAE, treatment-emergent adverse event.
1. Reich K, et al. Lancet. 2019;394:576–586.
SKYRIZI safety profile in Phase 3 IMMvent trial1
Overview of TEAEs reported in IMMvent Phase 3 trial from Week 16 to Week 441

| Re-randomized patients | |||||
|---|---|---|---|---|---|
| Events, % | SKYRIZI (Week 0-16) to SKYRIZI (Week 16-44; n=294) | ADA IR (Week 0-16) and PASI 50 to PASI <90 (Week 16) to SKYRIZI (Week 16-44; n=53) | ADA IR (Week 0-16) and PASI 50 to PASI <90 (Week 16) to ADA (Week 16-44; n=56) | ADA NR (Week 0-16) and PASI <50 (Week 16) to SKYRIZI (Week 16-44; n=38) | ADA (Week 0-16) and PASI >90 (Week 16) to ADA (Week 16-44; n=144) |
| Any adverse event | 188 (64%) | 40 (75%) | 37 (66%) | 23 (61%) | 98 (68%) |
| Drug-related adverse event* | 66 (22%) | 16 (30%) | 12 (21%) | 8 (21%) | 28 (19%) |
| Serious adverse event | 12 (4%) | 3 (6%) | 2 (4%) | 4 (11%) | 5 (3%) |
| Drug-related serious adverse event* | 4 (1%) | 1 (2%) | 0 | 2 (5%) | 1 (1%) |
| Severe adverse event | 14 (5%) | 3 (6%) | 5 (9%) | 4 (11%) | 6 (4%) |
| Adverse event leading to drug discontinuation | 5 (2%) | 0 | 3 (5%) | 1 (3%) | 1 (1%) |
| Infection | 119 (40%) | 25 (47%) | 18 (32%) | 15 (39%) | 68 (47%) |
| Serious infection | 3 (1%) | 2 (4%) | 0 | 2 (5%) | 2 (1%) |
| Opportunistic infection | 2 (1%) | 0 | 0 | 0 | 0 |
| Active tuberculosis | 0 | 0 | 0 | 0 | 0 |
| Latent tuberculosis† | 1(<1%) | 1 (2%)‡ | 0 | 1 (3%) | 0 |
| Serious hypersensitivity | 0 | 0 | 0 | 0 | 0 |
| Adjudicated major adverse cardiovascular event | 0 | 0 | 0 | 1 (3%)§ | 0 |
| Hepatic events | 8 (3%) | 1 (2%) | 4 (7%) | 0 | 6 (4%) |
| Malignancy | 2 (1%) | 0 | 0 | 0 | 0 |
| Malignancy excluding non-melanoma skin cancer | 1(<1%)¶ | 0 | 0 | 0 | 0 |
| Deaths (including non-treatment emergent) | 0 | 0 | 0 | 0 | 0 |
Data are n (%).
*Investigator assessed adverse event as possibly related to study drug. †Tuberculosis testing was done at screening and at the end of treatment using QuantiFERON or Purified Protein Derivative skin test. ‡One patient with mycobacterium tuberculosis complex tested positive during the study, without symptoms. §One patient with coronary artery occlusion adjudicated as type 1 myocardial infection on study day 202 (event was not considered to be study drug related by investigator). ¶One patient was diagnosed with prostate adenocarcinoma on study day 202 (event was not considered to be study drug related by the investigator).
ADA, adalimumab; AE, adverse event; IR, intermediate responder; NR, non-responder; PASI, Psoriasis Area and Severity Index; TEAE, treatment-emergent adverse event.
1. Reich K, et al. Lancet. 2019;394:576–586.
SKYRIZI showed a consistent, short-term safety profile across four Phase 3 psoriasis trials1-3
Incidence of TEAEs reported across the four pivotal Phase 3 trials across 1504 patients in the first 16 weeks of treatment1,2

| UltIMMa-11 (N=506) | UltIMMa-21 (N=491) | IMMvent2 (N=605) | IMMhance3 (N=507) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Events, % | SKYRIZI (n=304) | PBO (n=102) | UST (n=100) | SKYRIZI (n=294) | PBO (n=98) | UST (n=99) | SKYRIZI (n=301) | ADA (n=304) | SKYRIZI (n=407) | PBO (n=100) |
| Any AE | 49.7 | 51.0 | 50.0 | 45.6 | 45.9 | 53.5 | 56 | 57 | 45.7 | 49.0 |
| Serious AEs | 2.3 | 2.9 | 8.0 | 2.0 | 1.0 | 3.0 | 3.0 | 3.0 | 2.0 | 8.0 |
| Any adjudicated MACE | 0 | 0 | 0 | 0 | 0 | 0 | <1.0† | 0 | 0 | 1.0‡ |
| Any serious infection | 0.3 | 0 | 3.0 | 1.0 | 0 | 1.0 | <1.0 | 0.3 | 0.2 | 1.0§ |
| Any malignant tumor | 0.3 | 1.0 | 0 | 0.3 | 0 | 0 | <1.0 | 0.3 | 0.7‖ | 0 |
| Deaths (incl. non- treatment-emergent) | 0 | 0 | 0 | 0.3* | 0 | 0 | <1.0† | 0.7 | 0 | 0 |
*UltIMMa: One non–treatment-emergent death of unknown cause on study Day 189 that occurred 161 days after the last dose of study drug. †IMMvent: One patient with acute myocardial infarction on study Day 73 (event was not considered to be study drug related by investigator). ‡IMMhance: One patient with stroke reported as ischemic stroke on study Day 95. §IMMhance: One patient with esophageal carcinoma reported on study Day 16, with patient experiencing 40 lbs weight loss 6 months prior to study participation; one patient with malignant melanoma in situ reported on study Day 102, study drug was not interrupted; one patient with a cutaneous squamous cell carcinoma reported on study Day 89, study drug not interrupted. ‖IMMhance: Of the 31 patients with latent TB who did not receive prophylaxis during the study, none developed active TB during the mean follow-up of 55 weeks on SKYRIZI.
AE, adverse event; MACE, major adverse cardiac event; PBO, placebo; TB, tuberculosis; TEAE, treatment-emergent adverse event; UST, ustekinumab.
1. Gordon KB, et al. Lancet. 2018;392:650–61; 2. Reich K, et al. Lancet. 2019;394:576–86; 3. Blauvelt A, et al. JAMA Dermatol. 2020;156(6):649–58.
Rates of AEs of special interest up to 7.8 years in patients with psoriasis1,2*
Treatment-emergent events with SKYRIZI per 100 PY : Week 16 (five-study pool)1 and long-term (20-study pool)2 treatment exposure*

| Events, (E/100 PY) [95% CI] | Week 161 | Long-term all SKYRIZI2
(n=3502; 11,953.4 PY) |
|
|---|---|---|---|
| PBO (n=300; 92.0 PY) |
SKYRIZI 150 mg (n=1306; 402.2 PY) |
||
| Overall AEs | 261 (284) | 1,279 (320.0) | 17,620 (147.4) [145.2, 149.6] |
| SAEs | 16 (17.4) | 40 (9.9) | 891 (7.5) [7.0, 8.0] |
| AE leading to discontinuation | 9 (9.8) | 11 (2.7) | 225 (1.9) [1.6, 2.1] |
| AE leading to death | 0 | 1 (0.2) | 32 (0.3) [0.2, 0.4] |
| Serious infections | 1 (1.1) [<0.1, 6.1] | 7 (1.7) [0.7, 3.6] | 145 (1.2) [1.0, 1.4] |
| Malignant tumors excluding NMSC | 0 [0, 3.3] | 3 (0.7) [0.2, 2.2] | 74 (0.6) [0.5, 0.8] |
| NMSC | 1 (1.1) [<0.1, 6.1] | 3 (0.7) [0.2, 2.2] | 72 (0.6) [0.5, 0.8] |
| Adjudicated MACE | 1 (1.1) [<0.1, 6.1] | 1 (0.2) [<0.1, 1.4] | 62 (0.5) [0.4, 0.7] |
| Serious hypersensitivity | 0 | 0 | 7 (<0.1) [0.0, 0.1] |
| Depression | 2 (2.2) [0.3, 7.9] | 4 (1.0) [0.3, 2.6] | 75 (0.6) [0.5, 0.8] |
| Suicidal ideation and behavior | 1 (1.1) [<0.1, 6.1] | 2 (0.5) [<0.1, 1.8] | 8 (<0.1) [0.0, 0.1] |
Short-term safety through Week 16 was evaluated using data integrated from 5 Phase 2 and 3 trials in patients with moderate to severe plaque psoriasis: Trial 1311.2, UltIMMa-1, UltIMMa-2, IMMhance, and IMMvent. *Week 16 (five-study pool) and long-term (up to 7.8 years, 20-study pool) represent different pools of patients with varying lengths of treatment exposure included in the long-term set. SKYRIZI events counted in the Week 16 column are also included in the long-term column. In long-term safety, median treatment duration was 4.2 years (range 3 days to 7.8 years), ≥6 months in 3172 patients (90.6%), ≥1 year in 2680 patients (76.5%), ≥2 years in 2097 patients (59.9%), ≥3 years in 1960 patients (66.0%), ≥4 years in 1816 patients (51.9%), ≥5 years in 1266 patients (36.2%), and ≥6 years in 154 patients (4.4%).
AE, adverse event; E, event; MACE, major adverse cardiovascular event; NMSC, non-melanoma skin cancer; PBO, placebo; PY, patient-years; SAE, serious adverse event; TEAE, treatment-emergent adverse event.
1. Gordon KB, et al. Br J Dermatol. 2022;186(3):466–475; 2. Gordon KB, et al. Poster presented at: the 31st Congress of the European Academy of Dermatology and Venerology, 7–10 September 2022. P1607.
Ahmed achieved PASI 90 after 4 doses of SKYRIZI
Ahmed reached PASI 90 by Week 36
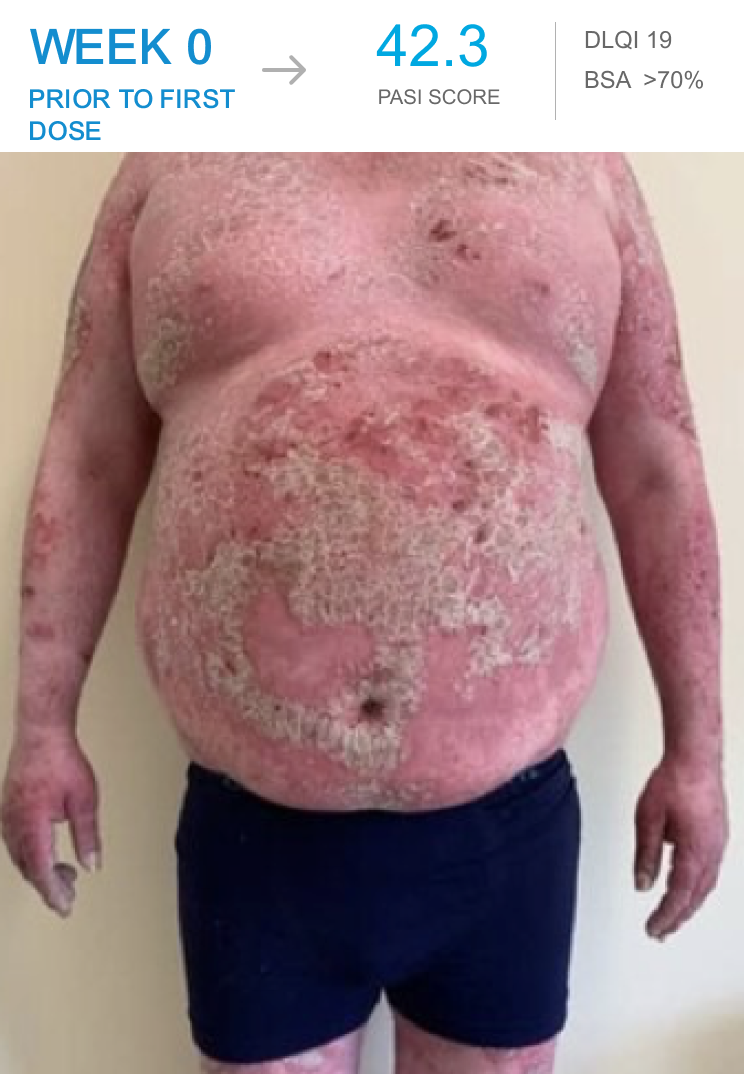
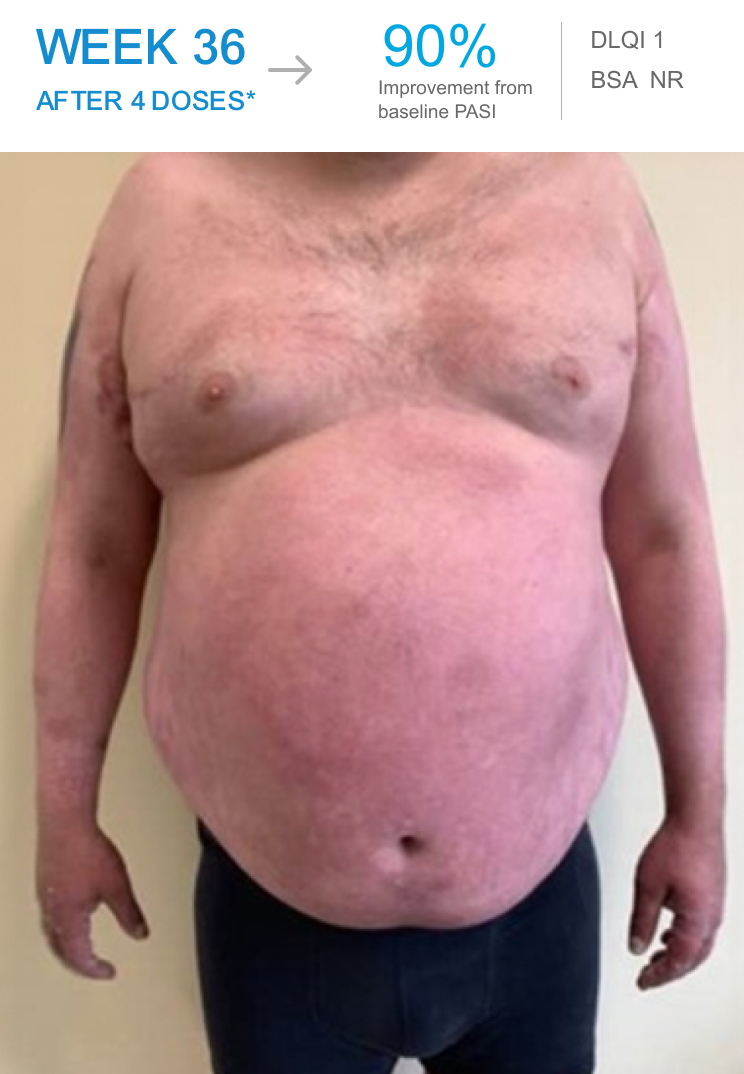

*Patient was started on SKYRIZI at a dose of 150 mg subcutaneously at Week 0, Week 4, and every 12 weeks thereafter.
BSA, body surface area; DLQI, Dermatology Life Quality Index; NR, not reported; PASI, Psoriasis Area and Severity Index; TEAE, treatment-emergent adverse event.
Deidentified patient case provided by Prof. Tiago Torres, Instituto de Ciências Biomédicas Abel Salazar, University of Porto, Porto, Portugal, with patient’s consent. For illustrative purposes only, individual results will vary depending on patient’s unique situation. Pseudonyms have been used to protect patient anonymity. Case study is a clinical observation of a single patient experience. AbbVie makes no representation or promise of similar results in any patient.
*Patient was started on SKYRIZI at a dose of 150 mg subcutaneously at Week 0, Week 4, and every 12 weeks thereafter.
DLQI, Dermatology Life Quality Index; PASI, Psoriasis Area and Severity Index.
Deidentified patient case provided by Prof. Tiago Torres, Instituto de Ciências Biomédicas Abel Salazar, University of Porto, Porto, Portugal, with patient’s consent. For illustrative purposes only, individual results will vary depending on patient’s unique situation. Pseudonyms have been used to protect patient anonymity. Case study is a clinical observation of a single patient experience. AbbVie makes no representation or promise of similar results in any patient.
1. Reich K, et al. Lancet. 2019;394:576–586, and Supplementary appendix.
Summary of Ahmed’s case
- Ahmed is a 38-year-old male with severe plaque-type psoriasis and history of inadequate response to adalimumab and methotrexate
- Comorbidities included obesity and hyperlipidemia
- Ahmed had a PASI score of 42.3 at baseline visit and achieved PASI 90 response by Week 36 after initiating SKYRIZI (risankizumab)
- A summary of the data for SKYRIZI shown in this case is included on the following page
PASI, Psoriasis Area and Severity Index.
Deidentified patient case provided by Prof. Tiago Torres, Instituto de Ciências Biomédicas Abel Salazar, University of Porto, Porto, Portugal, with patient’s consent. For illustrative purposes only, individual results will vary depending on patient’s unique situation. Pseudonyms have been used to protect patient anonymity. Case study is a clinical observation of a single patient experience. AbbVie makes no representation or promise of similar results in any patient.
1.SKYRIZI Summary of Product Characteristics. AbbVie Ltd; September 2023.
Summary of SKYRIZI data presented in this clinical case
Superior to adalimumab
At achieving complete skin clearance in PsO at Week 16 and Week 44: IMMvent pivotal trial (p<0.0001)1*
Week 16
- 40% of patients who received SKYRIZI achieved PASI 100 vs 23% of patients who received adalimumab (p <0.0001)
- 72% of patients who received SKYRIZI achieved PASI 90† vs 47% of patients who received adalimumab (p <0.0001)
Among adalimumab intermediate responders (PASI 50 to <PASI 90) who were re-randomized at Week 16:1
Week 44
- 40% of patients who received SKYRIZI achieved PASI 100 vs 7% of patients who received adalimumab (p<0.0001)
- 66% of patients who received SKYRIZI achieved PASI 90† vs 21% of patients who received adalimumab (p<0.0001)
Safety data from IMMvent pivotal trial‡
- Treatment-emergent adverse events were similar between SKYRIZI and adalimumab1
- No new safety signals were identified1
- Safety observed with SKYRIZI in IMMvent was consistent with the safety reported in three other SKYRIZI phase 3 trials (UltIMMa-1, UltIMMa-2, and IMMhance)1,2
Dosing simplicity
No more than 4 injections per year after initiation doses for moderate to severe psoriasis3§
- No dose adjustment required regardless of baseline characteristics, including BMI and weight3,4,5¶
- SKYRIZI is dosed 150 mg (one 150-mg subcutaneous injection) at Week 0, Week 4, and every 12 weeks thereafter3
- Consideration should be given to discontinuing treatment in patients who have shown no response after 16 weeks of treatment. Some plaque psoriasis patients with initial partial response may subsequently improve with continued treatment beyond 16 weeks3
*Co-primary endpoint. Note: some studies were designed with primary endpoints in two parts (Part A and Part B), per protocol. All other endpoints were ranked secondary. In IMMvent, PASI 90 and sPGA 0/1 (sPGA 0/1 data not shown) were co-primary endpoints. †Primary endpoint. All other endpoints were ranked secondary. ‡Integrated all-SKYRIZI safety data set from 20 completed or ongoing Phase 1–3 risankizumab clinical trials in plaque psoriasis (data cutoff March 25, 2022): UltIMMa-1, UltIMMa-2, IMMhance, IMMvent, Trials 1311.1, 1311.2, 1311.13, 1311.38, M15-999, M16-176, M16-177, M16-178, M16-766, M16-005, M16-007, M19-164, and LIMMitless. Median duration of treatment was 4.2 years (range 3 days to 7.8 years). §Maintenance dosing (one 150-mg subcutaneous injection/dose) every 12 weeks following a starter dose at Week 0 and Week 4. ¶ SKYRIZI clearance and volume of distribution increase as body weight increases which may result in reduced efficacy in subjects with high body weight (>130 kg). However, this observation is based on a limited number of subjects. No dose adjustment based on body weight is currently recommended.
BMI, body mass index; PASI, Psoriasis Area and Severity Index; PsO, psoriasis; sPGA, static Physician’s Global Assessment.
1. Reich K, et al. Lancet. 2019;394:576–586; 2. Gordon KB, et al. Poster presented at: the 31st Congress of the European Academy of Dermatology and Venerology, 7–10 September 2022. P1607; 3. SKYRIZI Summary of Product Characteristics. AbbVie Ltd; September 2023; 4. Strober et al. J Eur Acad Dermatol Venereol. 2020;34(12):2830-2838; 5. Strober B, et al. Poster presented at: Skin Inflammation & Psoriasis International Network (SPIN) 6th Congress; April 25–27, 2019; Paris, France. Poster P081.
EU Indication and Important Safety Information for SKYRIZI (risankizumab)1
INDICATION
SKYRIZI is indicated for the treatment of moderate to severe plaque psoriasis in adults who are candidates for systemic therapy.
SKYRIZI, alone or in combination with methotrexate (MTX), is indicated for the treatment of active psoriatic arthritis in adults who have had an inadequate response or who have been intolerant to one or more disease-modifying antirheumatic drugs (DMARDs).
IMPORTANT SAFETY INFORMATION
Risankizumab is contraindicated in patients hypersensitive to the active substance or to any of the excipients, and in patients with clinically important active infections (e.g. active tuberculosis). Risankizumab may increase the risk of infection. In patients with a chronic infection, a history of recurrent infection, or known risk factors for infection, risankizumab should be used with caution. Treatment with risankizumab should not be initiated in patients with any clinically important active infection until the infection resolves or is adequately treated.
Patients treated with risankizumab should be instructed to seek medical advice if signs or symptoms of clinically important chronic or acute infection occur. If a patient develops such an infection or is not responding to standard therapy for the infection, the patient should be closely monitored and risankizumab should not be administered until the infection resolves.
Prior to initiating treatment with risankizumab, patients should be evaluated for tuberculosis (TB) infection. Patients receiving risankizumab should be monitored for signs and symptoms of active TB. Anti-TB therapy should be considered prior to initiating risankizumab in patients with a past history of latent or active TB in whom an adequate course of treatment cannot be confirmed.
Prior to initiating therapy with risankizumab, completion of all appropriate immunizations should be considered according to current immunization guidelines. If a patient has received live vaccination (viral or bacterial), it is recommended to wait at least 4 weeks prior to starting treatment with risankizumab. Patients treated with risankizumab should not receive live vaccines during treatment and for at least 21 weeks after treatment.
If a serious hypersensitivity reaction occurs, administration of risankizumab should be discontinued immediately and appropriate therapy initiated.
The most frequently reported adverse reactions were upper respiratory infections (from 13% in psoriasis to 15.6% in Crohn's disease). Commonly (greater than or equal to 1/100 to less than 1/10) reported adverse reactions included tinea infections, headache, pruritus, rash, fatigue and injection site reactions.
This is not a complete summary of all safety information. Please see the SmPC for complete prescribing information.
1. SKYRIZI (risankizumab) Summary of Product Characteristics. AbbVie Ltd; September 2023.
EU Indication and Important Safety Information for HUMIRA (adalimumab)1
INDICATION
HUMIRA is indicated for the treatment of moderate to severe chronic plaque psoriasis in adult patients who are candidates for systemic therapy.
IMPORTANT SAFETY INFORMATION
HUMIRA is contraindicated in patients with active tuberculosis or other severe infections such as sepsis, and opportunistic infections and in patients with moderate to severe heart failure (New York Heart Association NYHA class III/IV). It is also contraindicated in patients hypersensitive to the active substance or to any of the excipients; serious allergic reactions including anaphylaxis have been reported.
The use of HUMIRA increases the risk of developing serious infections, including hepatitis B reactivation, which may, in rare cases, be life-threatening. Rare cases of lymphoma and leukemia have been reported in patients treated with HUMIRA.
On rare occasions, a severe type of cancer called hepatosplenic T-cell lymphoma has been observed and often results in death. A risk for the development of malignancies in patients treated with tumor necrosis factor-antagonists cannot be excluded. Rare cases of pancytopenia, aplastic anemia, demyelinating disease, lupus, lupus-related conditions and Stevens-Johnson syndrome have been reported in patients treated with HUMIRA.
The most frequently reported adverse events across all indications included respiratory infections, injection site reactions, headache and musculoskeletal pain.
This is not a complete summary of all safety information. Please see the product labeling for complete safety information.
1. HUMIRA (adalimumab) Summary of Product Characteristics. AbbVie Ltd; October 2022.

















